Decoding the Synergistic Lethality of Heavy Metal Co-Exposure
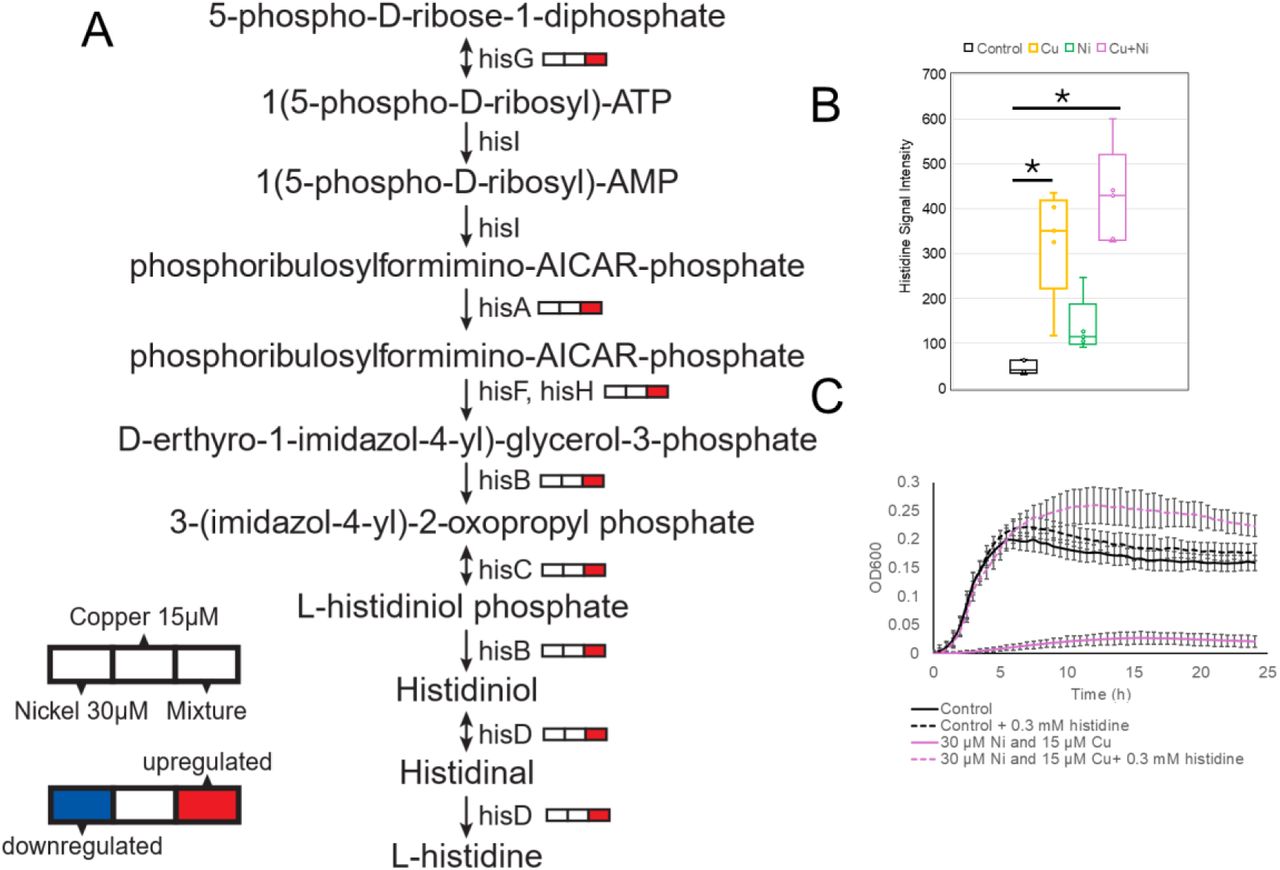
Pollutants rarely exist in isolation in the environment. The research team observed that co-exposure to Nickel (Ni) and Copper (Cu) caused a synergistic lethality (1+1 > 2). While transcriptomic analysis (Figure 2 in the study) indicated broad gene expression changes, the researchers required rigorous metabolic validation to locate the actual "synergistic blockade" causing cellular death.
We deployed an Untargeted Metabolomics Profiling (Bundle A) workflow to contrast control, single-exposure, and co-exposure groups. By using high-resolution mass spectrometry, we captured intracellular metabolite alterations and applied advanced chemometrics to filter out generalized stress markers, focusing exclusively on the lipid and metabolite species uniquely depleted in the synergistic group.
The mass spectrometry data revealed that Ni and Cu do not just stack damage; they synergistically obstruct specific synthesis hubs (such as sulfur assimilation and histidine biosynthesis), obliterating the cell's ability to maintain structural integrity. This precise biochemical validation provided the irrefutable evidence chain needed for establishing multi-metal exposure limits.