
- Home
- Services
- (Untargeted) Lipidomics Profiling
- Mammals Untargeted Lipidomics
- Brain Lipidomics Analysis
- Mouse Lipidomics
- Platelet Lipidomics
- Tissues Lipidomics
- Urinary Lipidomics
- Biofluid Lipidomics
- Mitochondria Lipid Metabolism Analysis
- Skin Lipidomics
- Cells Lipidomics
- Conditioned Medium Lipidomics
- Exosomes Lipidomics
- Plasma Lipidomics
- Serum Lipidomics Analysis
- Milk Lipidomics
- Microorganisms Untargeted Lipidomics
- Plant Lipidomics Analysis
- Algae Lipidomics Analysis
- Foods Untargeted Lipidomics
- Mammals Untargeted Lipidomics
- Targeted Lipidomics
- Fatty Acyls
- Acylcarnitine
- Hepoxilins
- Resolvins
- Isoprostanes
- Protectins D1
- Maresins
- Fatty Acid
- Fatty Acid Oxidation and its Metabolites
- Fatty Acid Methyl Ester
- Free Fatty Acids
- Short Chain Fatty Acids
- Volatile Fatty Acid (VFA)
- Fatty Acid Esters of Hydroxy Fatty Acids (FAHFAs)
- Oxylipins
- Phospholipid Fatty Acids
- Prostaglandins
- Leukotrienes
- Thromboxanes
- Lipoxins
- Endocannabinoids
- Hydroxy-Eicosatetraenoic Acids
- Mycolic Acids
- Epoxyeicosatrienoic Acids
- Neurofurans
- Jasmonic Acids
- Sophorolipids
- Rhamnolipids
- Isofurans
- Phytoprostanes
- Eicosanoid
- Wax Esters
- Heptadecanoids
- Glycerophospholipids
- Phosphatidylcholine
- Phosphatidylethanolamine
- Phosphatidylserine
- Phosphatidic Acid
- Phosphatidylglycerol
- Phosphatidylinositol
- Phosphoinositides
- Cardiolipins
- Lysophosphatidic Acid
- Lysophosphatidylcholine
- Lysophosphatidylserine
- Lysophosphatidylglycerol
- Lysophosphatidylethanolamine
- Lysophosphatidylinositol
- Ether-linked Phosphatidylcholine
- Plasmalogen
- Lysophospholipid Analysis Service
- Ether-linked Phosphatidylethanolamine
- CDP-Diacylglycerols
- LysoPAF
- LPE O-
- Bis(monoacylglycero)phosphate (BMP/LBPA)
- Glycerolipids
- Sphingolipids
- Glycolipids
- Acylceramides
- Ceramide 1-Phosphates
- Ceramides
- Cerebroside
- Dihexosylceramides
- Ganglioside
- Globoside
- Glucocerebroside
- Glucosylsphingosine
- Glycosphingolipid
- Hexosylceramide
- Inositol-P-Ceramide
- Phytoceramide
- Sphingomyelins
- Sphingosine 1-phosphate
- Sphingosine Base
- Sulfatides
- Lactosylceramide
- Glucosylceramide
- Sterol Lipids
- Prenol Lipids
- Phospholipids
- Saccharolipids
- Polyketides
- Fatty Acyls
- Lipidomics Bioinformation Analysis
- MALDI-Imaging Lipidomic Service
- Metabolic Flux Analysis Services
- Others Service
- (Untargeted) Lipidomics Profiling
- Applications
- Pharma & Biotech
- Food & Nutrition
- Disease Research
- Precision Oncology & Ferroptosis Lipidomics
- Neuro-Metabolism Lipidomics for Brain Health Research
- Dermatology Lipidomics Services for Skin Barrier Function
- Immunology Lipidomics Services for Inflammatory Signaling
- Lipidomics in MAFLD, NASH and Cardiovascular Research
- Lipidomics for Immune Disease Research
- Environmental Toxicology
- Agriculture
- Microorganism
- Drug Analysis
- Fermentation Conditions Optimization Analysis
- Microbial Lipidomics for Microbial Cell Factory Engineering
- Antimicrobial Resistance and Bacterial Membrane Biology Lipidomics Service
- Fermentation Process Optimization and Dynamic Monitoring Lipidomics
- Host-Pathogen Interaction Lipidomics Services
- Cosmetics & Personal Care
- Resource
- About Lipids
- Platform
- Service Flow
- Sample Guidelines
- Knowledge Center
- Applications of Lipidomics in Discovery of Disease Biomarkers
- Applications of Lipidomics in Food Quality and Safety
- Applications of Lipidomics in Nutrition and Health
- Biological Functions of Lipids and Abnormalities of Lipid Metabolism
- Function, Application and Detection of Plant Sterols
- How to Detect Steroid Hormones?
- Lipidomics and Cancer Research
- Lipidomics in Microbiology
- Metabolic Flux Analysis in Tumor Research
- Detection of Intracellular Ceramide
- The Role of Mass Spectrometry and Chromatography in Lipidomics
- Untargeted vs. Targeted Lipidomics—Understanding the Differences
- Exploring the Roles of Phosphatidic Acid in Lipid Metabolism and Lipidomics Research
- Fatty Acids and Fatty Acid Metabolism
- Nomenclature and Function of Ceramide and Diseases Caused by Abnormal Ceramide Metabolism
- Lipoprotein A in Cardiovascular Disease
- The Role of Short-Chain Fatty Acids in Metabolic Diseases
- Overview of Long Chain Fatty Acid
- Overview of Omega-6 Fatty Acids
- Overview of Phosphatidylcholine
- Overview of Phosphatidylethanolamine
- Overview of Phosphatidylglycerol
- Lipidomics Databases and Software Tools for Biomedical Research
- What is Untargeted Lipidomics?
- Common Lipidomics Databases and Software
- What is Targeted Lipidomics?
- Overview of Gas Chromatography-Mass Spectrometry (GC-MS)
- Sphingosine-1-Phosphate: Structure, Functions, and Role in Cancer
- Glycosphingolipids: Structure, Metabolism, Functions, Analytical Methods and Biological Significance
- Phosphatidylserine Metabolism Structure, Functions, and Analytical Techniques
- Overview of Medium Chain Fatty Acids (MCFAs)
- Overview of Short-Chain Fatty Acids
- Short-Chain Fatty Acids in Gut Health and Metabolism
- Comprehensive Overview of Omega-3 Fatty Acids
- Difference Between Omega-3 and Omega-6 Fatty Acid
- Omega-3 Fatty Acids for Dogs & Cats
- Role of Omega-3 Fatty Acids in Brain Health
- Phospholipid Analysis Techniques: Principles, Methods, and Applications
- FAME Analysis Guide: Workflow, Sample Prep, Platform Selection, and QC Tips
- Arachidonic Acid Analysis: From Pathway Biology to Study Design and LC-MS/MS Assay Development
- How to Choose the Right Arachidonic Acid Analysis Strategy: LC-MS/MS, GC-MS, ELISA, and Panel Design
- When to Use Arachidonic Acid and Eicosanoid Analysis in Disease Research
- Advanced Arachidonic Acid Lipidomics and Multi-Omics Strategies for Mechanism and Preclinical Research
- Sample Preparation and Quality Control for Arachidonic Acid and Eicosanoid LC-MS/MS Results
- Saturated vs Unsaturated Fatty Acids: How to Select GC-FID, GC-MS, or LC-MS/MS for Quantitation
- Acid Sphingomyelinase Deficiency: Pathways, Sphingomyelinase Disorders, and Research Models
- How Saturated Fatty Acid Structure Affects GC Separation and Quantitative Accuracy
- Lecithin–Sphingomyelin Ratio (L/S Ratio): Biological Meaning and Research Applications
- Sphingomyelin Assays Compared: ELISA and Colorimetric Kits vs LC–MS/MS Quantification
- Neurolipidomics Strategy for Sphingomyelin Analysis in Brain Tissue and CSF
- Ceramide vs Sphingomyelin: Pathways, Differences & Measurement
- Sphingomyelin vs. Phosphatidylcholine: Structure, Composition, and Key Biological Differences
- Prostaglandin Measurement: LC-MS/MS vs ELISA—Choosing the Right Method
- COX-1 vs COX-2 Pathway: Mechanistic Differences and Analytical Strategies
- How to Prepare Samples for Prostaglandin Measurement
- 5 Key Research Areas Where Prostaglandin Analysis Is Transforming Biomedical Discovery
- How to Interpret Your Prostaglandin Analysis Report — From LC-MS/MS Data to Biological Insights
- Shotgun Lipidomics—Principles, Applications, and Best Practices
- The Role and Detection Methods of Long-Chain Fatty Acids
- How Are Short-Chain Fatty Acids Measured?
- Detection of Sphingomyelins
- Detection Strategies for Glycosphingolipids: Methods and Techniques
- Function and Analysis of Phosphatidic Acid
- Function and Analysis of Phosphatidylserine
- Function and Detection of Leukotrienes
- Phosphatidylcholine: Function and Detection
- Function and Detection of Sphingolipids
- Functions of Gangliosides and Association with Brain Diseases
- Metabolism and Detection of Sphingosine-1-Phosphate
- Metabolism and Detection of Triacylglycerol
- Phospholipids: Structure, Biosynthesis, Functions, and Role in Cellular Processes
- Analytical Methods to Quantify Free Fatty Acids
- Application of Lipidomics in Environmental Research
- Application of Lipidomics in Pharmaceutical Research
- Function and Detection Methods of Polyunsaturated Fatty Acids
- What Are Prostaglandins? A Guide to Function, Synthesis, and Measurement
- Introduction to Saturated Fatty Acids
- Methods of Lipidomics Analysis
- Infographic
- Brochures
- Blog
- Exosome Lipidomics: Understanding the Role of Lipids in Exosome Therapies
- Free Fatty Acids: Structure, Metabolism, Functions, and Measurement
- Understanding Plasma Lipidomics: An Introduction
- What is Thromboxane?
- What is Fatty Acid Methyl Ester?
- What is Triacylglycerol?
- What is Sterol?
- What is Sphingosine?
- What are Eicosanoids?
- What is GM2 Ganglioside?
- What is GM3 Ganglioside?
- What is Lipoxin?
- What is Monoacylglycerol?
- Medium Chain Fatty Acids: Metabolism, Health Benefits, and Analytical Methods
- What is Cardiolipin?
- What is Globoside?
- Glycerophospholipids: Essential Components of Cell Membranes and Cellular Functions
- What is Lipoprotein?
- Bile Acid: A Comprehensive Exploration of Function and Metabolism
- Exploring the Intricate Link Between Bile Acids and Disease
- Natural Lipids: Structure, Function, and Applications
- Waxes: Properties, Functions, and Analysis Techniques
- Cerebrosides: Structure, Function, and Analytical Methods
- Diacylglycerol: Structure, Functions, and Analytical Methods
- Lipidomics Pathway Analysis: Unraveling Cellular Lipid Metabolism and Signaling
- Company
- Inquiry

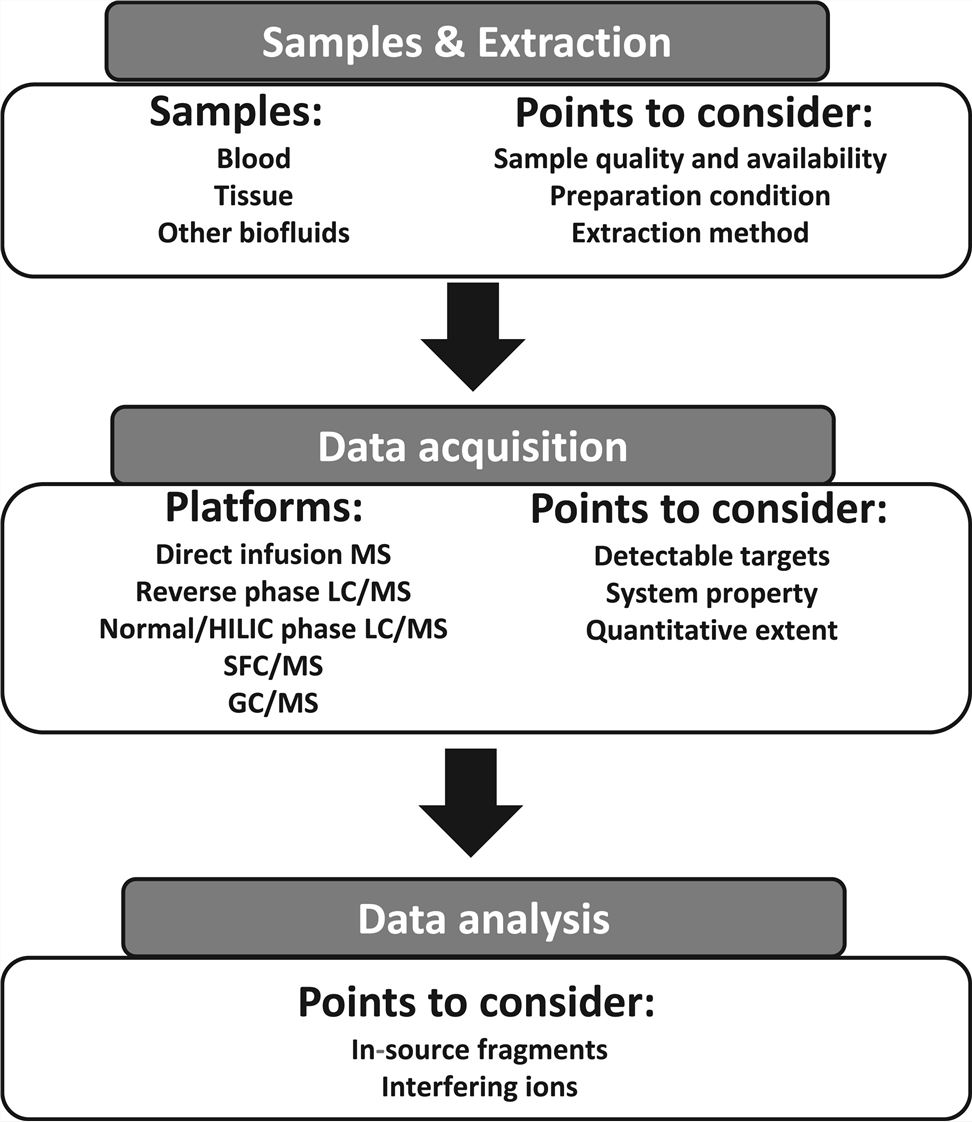 Fig1. Schematic workflow and points to consider in ADR lipidomics (Saito, Kosuke, 2020)
Fig1. Schematic workflow and points to consider in ADR lipidomics (Saito, Kosuke, 2020)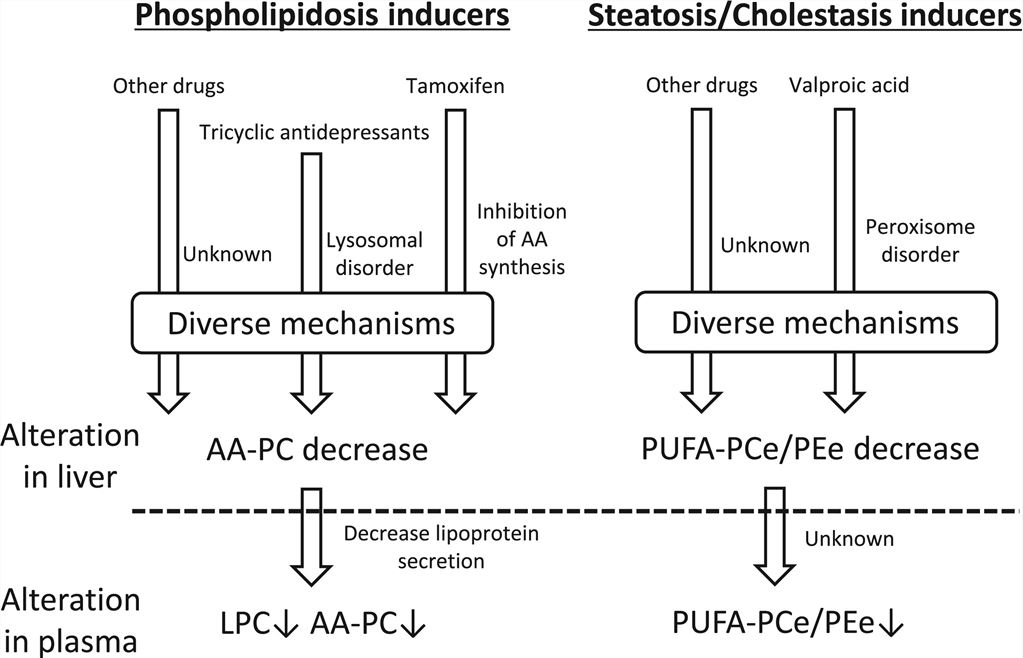 Fig2. Putative mechanisms underlying blood markers for lipid disorders in the liver biomarkers (Saito, Kosuke, 2020)
Fig2. Putative mechanisms underlying blood markers for lipid disorders in the liver biomarkers (Saito, Kosuke, 2020)